Methyl Green
Name: Methyl Green
CAS Registry Number: 7114-03-6
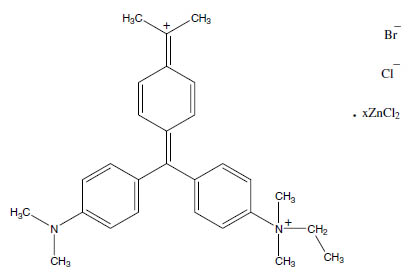
CA Index Name: Benzenaminium, 4-[[4-(dimethylamino)phenyl][4-(dimethyliminio)-2,5-cyclohexadien-1-yli-dene]methyl]-N-ethyl-N,N-dimethyl-, bromide chloride, compd. with zinc chloride (ZnCl2) (1:1:1:?)
Other Names: Ammonium, [α-[4-(dimethylamino)phenyl]-α-[p-(dimethyliminio)-2,5-cyclohexadien-1-yli-dene]-p-tolyl]ethyldimethyl-, bromide chloride, compd. with zinc chloride; Benzenaminium, 4-[[4-(dimethylami-no)phenyl][4-(dimethyliminio)-2,5-clohexadien-1-yli-dene]methyl]-N-ethyl-N,N-dimethyl-, bromide chloride, compd. with zinc chloride; Benzenaminium, 4-[[4-(di-methylamino)phenyl][4-(dimethyliminio)-2,5-cyclohex-adien-1-ylidene]methyl]-N-ethyl-N,N-dimethyl-, bromide chloride, compd. with zinc chloride (ZnCl2); Zinc chloride (ZnCl2), compd. with4-[[4-(dimethylamino)phe-nyl][4-(dimethyliminio)-2,5-cyclohexadien-1-ylidene] methyl]-N-ethyl-N,N-dimethylbenzenaminium bromide chloride; Ethyl green; Methyl green; Sigma Ethyl green; Sigma Methyl green; C. I. 42590
Merck Index Number: Not listed
Chemical / Dye Class: Triphenylmethane
Molecular Formula: C27H35BrClN3 × ZnCl2
Molecular Weight: 653.24
Physical Form: Red-brown powder
Solubility: Soluble in water, ethanol; insoluble in xylene
Melting Point: > 300 °C
pH Range: 0.1-2.3
Color Change at pH: Yellow (0.1) to greenish-blue (2.3)
Absorption (λmax): 629 nm, 423 nm
Synthesis: Synthetic method
Staining Applications: Brain; spinal cord; bacteria; chromosomes; nicotinic acetylcholine receptor; nucleic acids; phosphates; polyphosphates; tocopherol; tocopherol acetate
Biological Applications: Detecting phosphates, polyphosphates, tocopherol, tocopherol acetate
Industrial Applications: Spatial light modulators; security applications
Safety / Toxicity: No data available
Certification / Approval: Certified by Biological Stain Commission (BSC)
 Введение в прикладную энзимологию
Введение в прикладную энзимологию В учебном пособии освещены проблемы, связанные с созданием и внедрением нового ...
The texlbook is based on modern organic chemistry and considers the structure and chemical transformations of organic compounds, especially those that ...
 Гетероциклические соединения (комплект из 4 книг)
Гетероциклические соединения (комплект из 4 книг) Химия гетероциклических соединений в результате быстрого развития за последние ...
 Органическая химия. Углублённый курс. Том 2
Органическая химия. Углублённый курс. Том 2 Второй том систематизированного учебного и справочного руководства по ...